TAPSYSTEM Model 2A Maintenance
Proper operation of all biomedical equipment should be verified prior to each use.
Verification Procedure:
Equipment required
- Oscilloscope, Tektronix Model 2236 or equivalent
- Counter timer Fluke Model 1953A or equivalent
- 2 "fresh" D-size alkaline batteries (V > 1.4 volts)
- Test Load = 500 ohms, 1% resistor
- "Mini-grabber" test leads or equivalent
Setup
- Verify that the TAPSYSTEM Model 2A is turned off.
- Verify battery voltages and install within the unit.
- Connect the gray interface cable to the 2A.
- Connect a 500 ohm +/- 1% resistor load across the 2 pins at end of cable using test leads.
- Connect the oscilloscope and frequency counter across the load.
Note: Ground lead of the test equipment should be connected to the right hand side of the load resistor when looking into the interface cable end with its notch upward.
Procedure
- If stimulator output does not fall within the following guidelines, contact Customer Service.
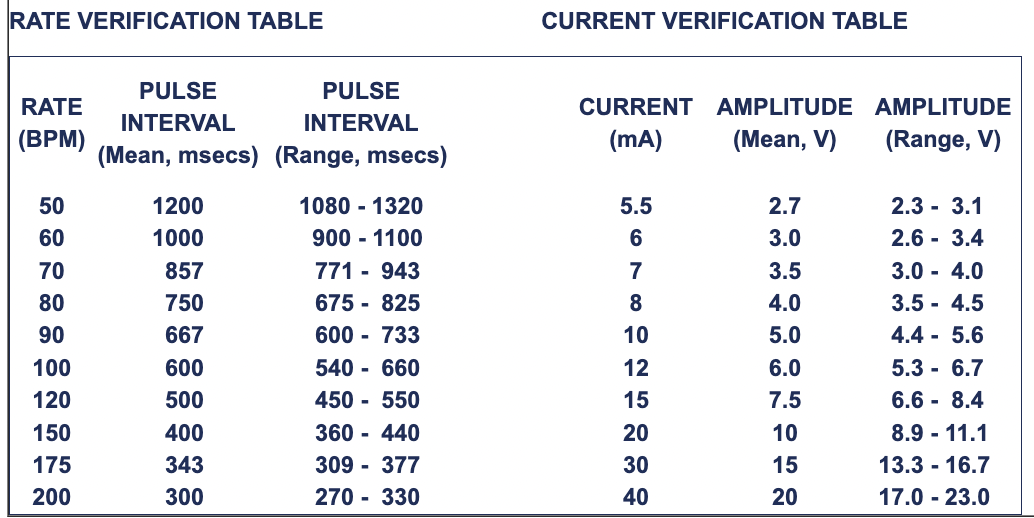
- Set panel controls as: CURRENT = 40 mA and RATE = 100 BPM (beats per minute).
- Pulse width check: Verify that output pulse across the load is 10 +/- 10% milliseconds at Vbatt>= 2.8Vdc.
- RATE and CURRENT verification: Vary the RATE and CURRENT as indicated in the Table below and verify that the pulse intervals (msecs) and amplitudes (volts) are as listed:
Trouble Shooting and Repairs:
The TAPSYSTEM Model 2A contains solid state devices. These circuits are susceptible to serious internal damage by the discharge of static electricity. To avoid damage, assure the proper grounding of tools, equipment and personnel. TAPSYSTEM Model 2A is to be serviced by CardioCommand only. To request repair service, contact Customer Service.
- Adjust the TAPSCOPE insertion depth.
- Increase CURRENT setting.
- Verify battery power.
- Check all connections.
- Monitor electrocardiogram for signs of A.V. Block.
Warranty
For a period of one year, CardioCommand warrants that the TAPSYSTEM Model 2A, when used in accordance with manufacturer's directions, is fit for the purpose and indications described in the labeling. Unless the products are used in accordance with directions, this warranty, and all other expressed or implied warranties, are void and of no effect. The manufacturer's sole obligation under this warranty shall be, at its option, to repair or replace the product. In no event shall the manufacturer be liable for incidental or consequential damages.
